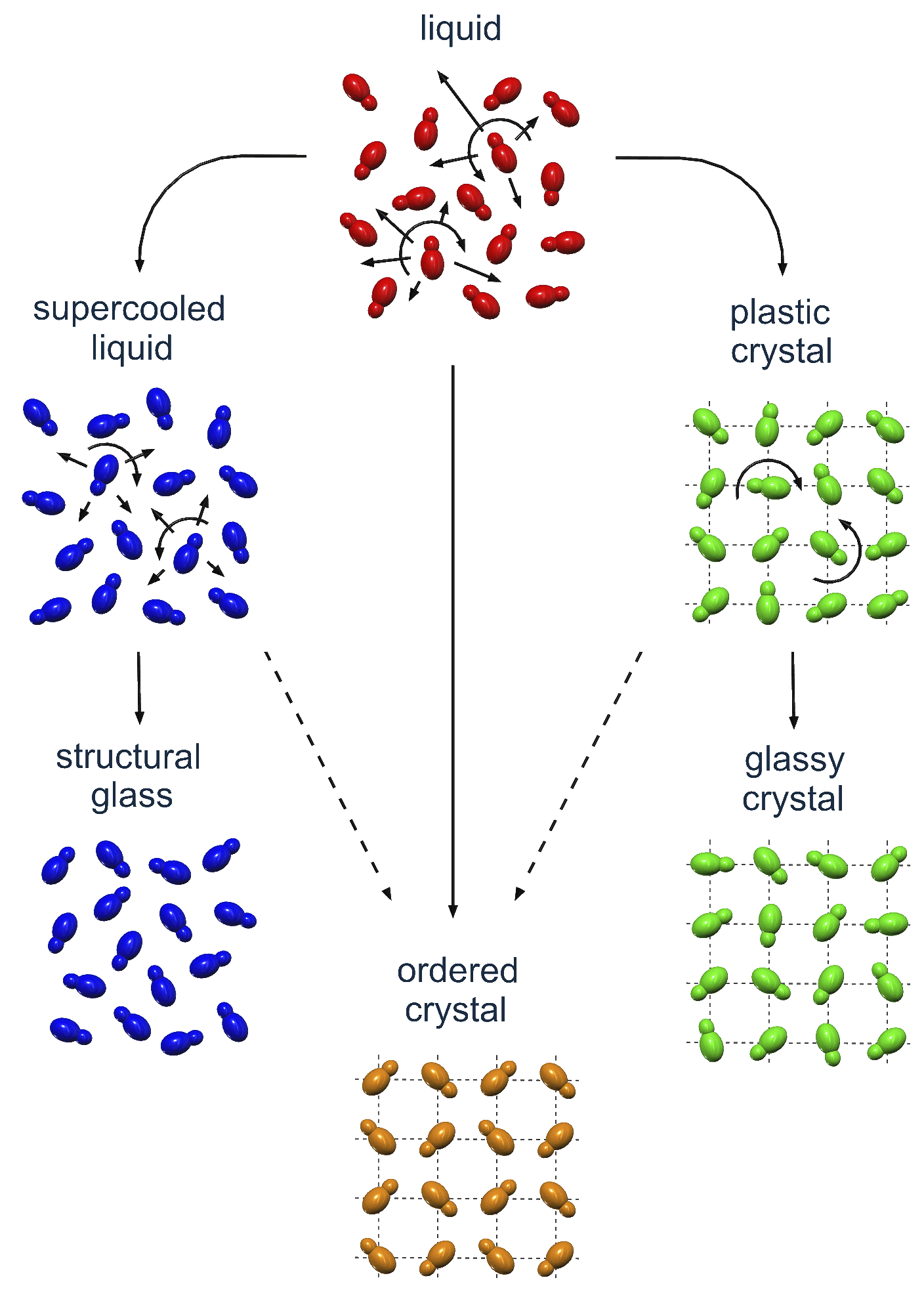
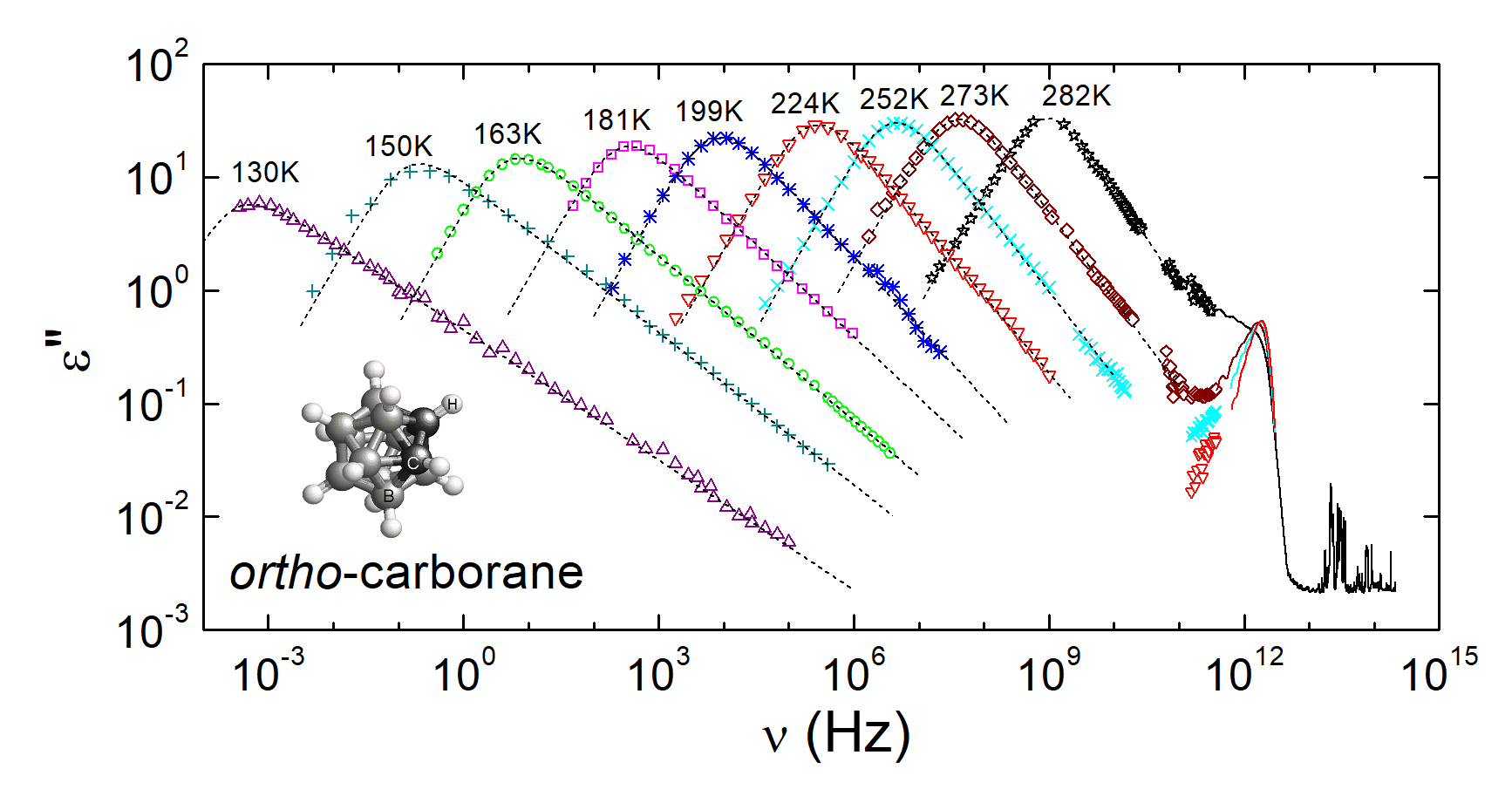
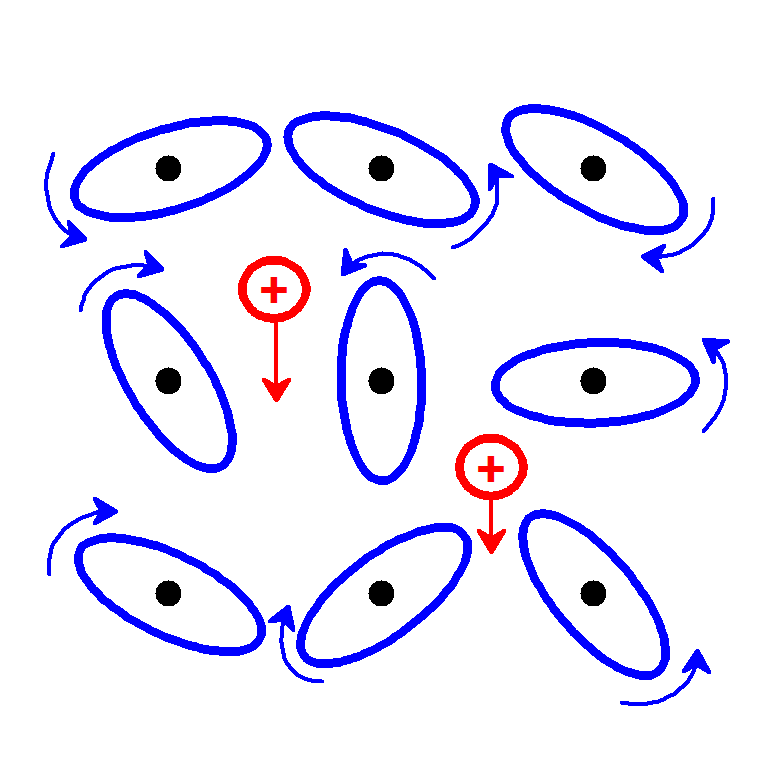
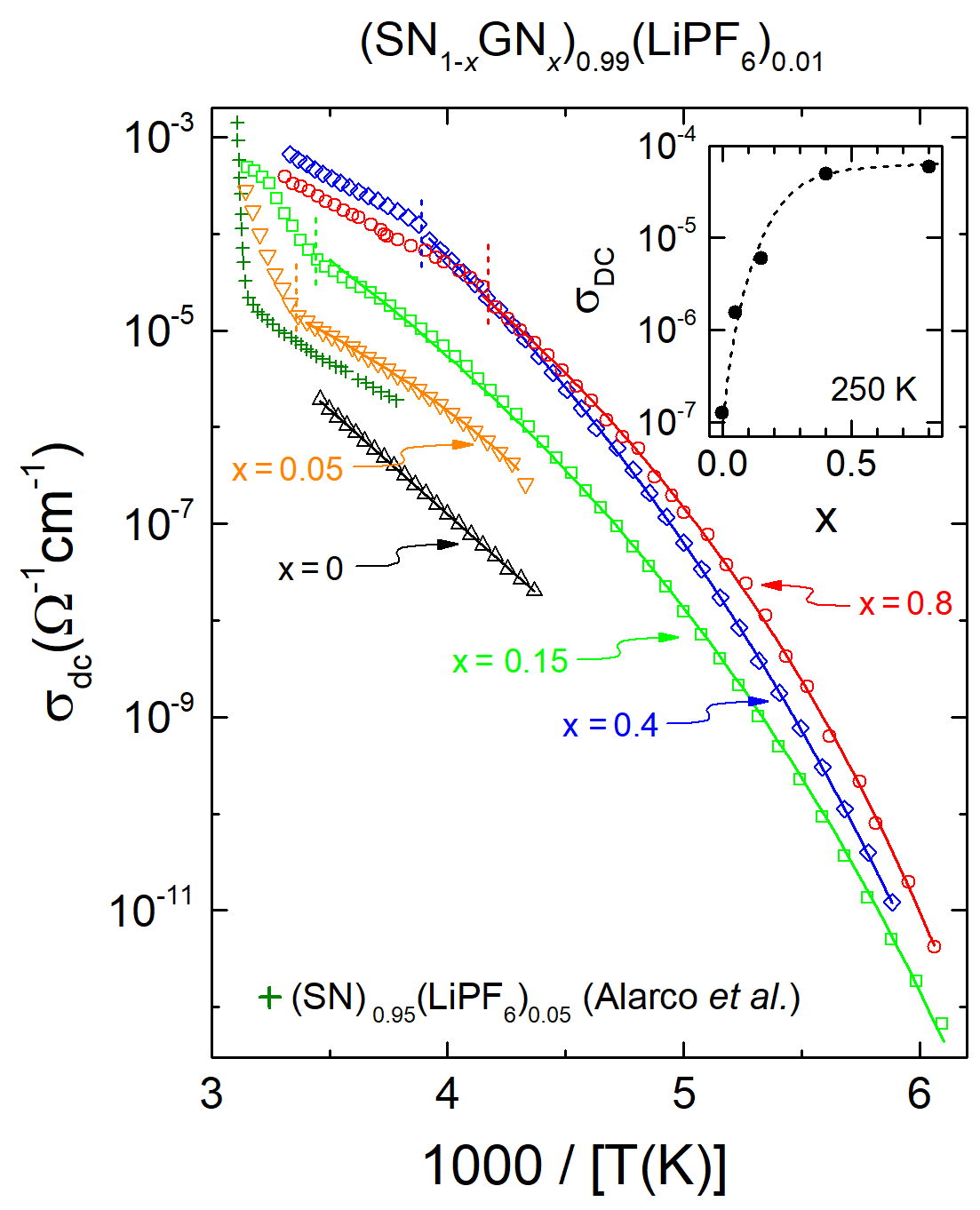
| [1] |
Molecular reorientation in ortho-carborane studied by dielectric spectroscopy
P. Lunkenheimer and A. Loidl,
J. Chem. Phys. 104, 4324 (1996).
[PDF]
|
| [2] |
Relaxations and fast dynamics of the plastic crystal cyclo-octanol investigated by broadband dielectric
spectroscopy
R. Brand, P. Lunkenheimer, and A. Loidl,
Phys. Rev. B 56, R5713 (1997).
[PDF]
|
| [3] |
Is there an excess wing in the dielectric loss of plastic crystals?
R. Brand, P. Lunkenheimer, U. Schneider, and A. Loidl,
Phys. Rev. Lett. 82, 1951 (1999).
[PDF]
|
| [4] |
α- and β-relaxation dynamics of a fragile plastic crystal
L.C. Pardo, P. Lunkenheimer, and A. Loidl,
J. Chem. Phys. 124, 124911 (2006).
[PDF]
|
| [5] |
High-frequency excitations in glassy crystals
P. Lunkenheimer and A. Loidl,
J. Non-Cryst. Solids 352, 4556 (2006). |
| [6] |
Relaxation dynamics and ionic conductivity in a fragile plastic crystal
Th. Bauer, M. Köhler, P. Lunkenheimer, A. Loidl, and C.A. Angell,
J. Chem. Phys. 133, 144509 (2010).
[PDF]
|
| [7] |
Supercooled-liquid and plastic-crystalline state in succinonitrile-glutaronitrile mixtures
M. Götz, Th. Bauer, P. Lunkenheimer, A. Loidl,
J. Chem. Phys. 140, 094504 (2014).
[PDF]
|
| [8] |
Cooperativity and heterogeneity in plastic crystals studied by nonlinear dielectric spectroscopy
M. Michl, Th. Bauer, P. Lunkenheimer, and A. Loidl,
Phys. Rev. Lett. 114, 067601 (2015).
[PDF]
|
| [9] |
Communication: Conductivity enhancement in plastic-crystalline solid-state electrolytes
K. Geirhos, P. Lunkenheimer, M. Michl, D. Reuter, and A. Loidl,
J. Chem. Phys. 143, 081101 (2015).
[PDF]
|
| [10] |
Nonlinear dielectric spectroscopy in a fragile plastic crystal
M. Michl, Th. Bauer, P. Lunkenheimer, and A. Loidl,
J. Chem. Phys. 144, 114506 (2016).
[PDF]
|
| [11] |
Variation of ionic conductivity in a plastic-crystalline mixture
D. Reuter, C. Geiß, P. Lunkenheimer, and A. Loidl,
J. Chem. Phys. 147, 104502 (2017).
[PDF]
|
| [12] |
Plastic-crystalline solid-state electrolytes: Ionic conductivity and orientational dynamics in nitrile
mixtures
D. Reuter, P. Lunkenheimer, and A. Loidl,
J. Chem. Phys. 150, 244507 (2019).
[PDF]
|
| [13] |
Ionic conductivity and relaxation dynamics in plastic crystals with nearly globular molecules
D. Reuter, K. Seitz, P. Lunkenheimer, and A. Loidl,
J. Chem. Phys. 153, 014502 (2020).
[PDF]
|
| [14] |
Dipolar relaxation, conductivity, and polar order in AgCN
P. Lunkenheimer, A. Loidl, and G. P. Johari,
J. Chem. Phys. 158, 184503 (2023).
|
| [15] |
Coupling and decoupling of molecular reorientation and charge transport
in Li-salt-doped cycloalcohol ion conductors
S. Lansab, Y. Hinz, B. Grabe, P. Lunkenheimer, and R. Böhmer,
Phys. Chem. Chem. Phys. 27, 16161 (2025).
[PDF]
|