THERMAL EXPANSION AND THE GLASS TRANSITION
THERMAL EXPANSION AND THE GLASS TRANSITION |

|
1. Introduction
The strong slowing down of particle motions, observed in glassforming liquids when approaching the glass-transition temperature Tg, makes the material fall out of thermodynamic equilibrium below Tg. Then the particles (atoms, molecules, ...) freeze into disordered positions forming a solid glass. Several models try to explain the many unusual phenomena at the glass transition by a "hidden" phase transition below Tg. This is accompanied by an increasing cooperativity of the particle motions (i.e., they do not move independently) upon cooling towards Tg, which was, e.g., confirmed by nonlinear dielectric measurements.
In contrast to the glass transition, the solid-liquid transition of crystalline materials is much better understood. In textbooks it is often explained in terms of the so-called Lindemann criterion: Upon heating, the thermal vibrations of the atoms or molecules become such vigorous that they break free from their crystalline, lattice-like arrangement and the material melts. Could such a simple picture also be relevant for the glass transition?
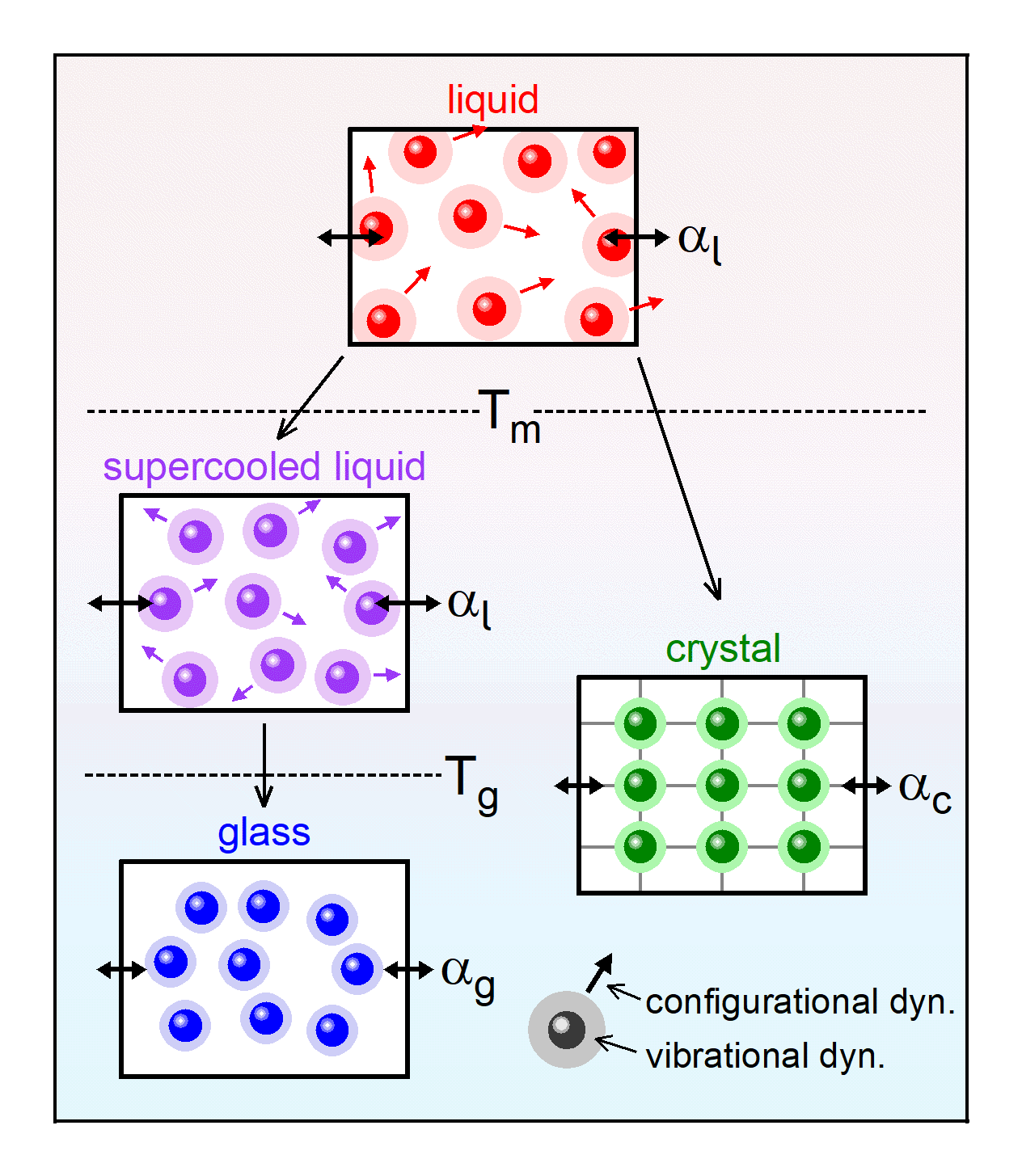
|
Fig. 1: Schematic presentation of the different contributions to the thermal expansion of liquids, glasses and crystals:
Local vibrational motions of the atoms (or molecules) are the dominant contribution to the thermal expansion in the
solid (glass and crystalline) states. The vibrational dynamics is indicated by the shaded areas around the
spheres, representing the atoms or molecules of the material. The higher temperatures in the liquid states
give rise to stronger vibrations, but they also enable additional translational ("configurational") motions, which further
enhance the thermal expansion (single-headed arrows). The double-headed arrows illustrate the resulting thermal
expansion. Tm is the melting and Tg the glass-transition temperature.
[from: P. Lunkenheimer, A. Loidl, B. Riechers, A. Zaccone, and K. Samwer, Thermal expansion and the glass transition, Nature Phys. 19, 694 (2023)]. |
To clarify this, we have analysed the thermal expansion coefficients α and the glass-transition temperatures Tg of more than 200 glasses and liquids, published during the past 100 years and belonging to very different material classes like conventional silicate glasses, molecular, ionic and metallic glasses and polymers. This is of interest because the mentioned thermal vibrations are also responsible for the thermal expansion of solid materials (see Fig. 1). If the basic ideas behind the Lindemann criterion are correct, the latter should be stronger for materials with lower melting temperatures, leading to an inverse proportionality of both quantities. Indeed, this is considered as well fulfilled for crystalline materials.
2. Universalities
Our analysis reveals, however, that an analogous inverse proportionality of thermal expansion and glass-transition temperature does not exist, implying that the Lindemann criterion is invalid for the glass transition (open symbols in Fig. 2b). However, both quantities are indeed correlated, but α depends more strongly on Tg (dashed line). It seems reasonable to ascribe this qualitatively different behaviour to the typical cooperativity of the particle dynamics in glass-forming materials.
The degree of cooperativity is different for each material and can be quantified by the so-called fragility index m, which can be determined, e.g., from the temperature dependence of the α relaxation. Interestingly, when we divide the thermal expansion coefficients of the different glasses by their fragility indices, glass-forming materials also exhibit an inverse proportionality of this scaled quantity with the glass-transition temperature (closed symbols in Fig. 2b). While vibrations also play a role for the glass transition, this finding evidences a significant additional influence of cooperativity. One can rationalize this as follows: For a glass to become liquid, it is not sufficient to simply overcome the interparticle binding energies like in a simple melting process. Instead, more energy has to be invested to break up the typical cooperative particle network that is common to glassy materials.
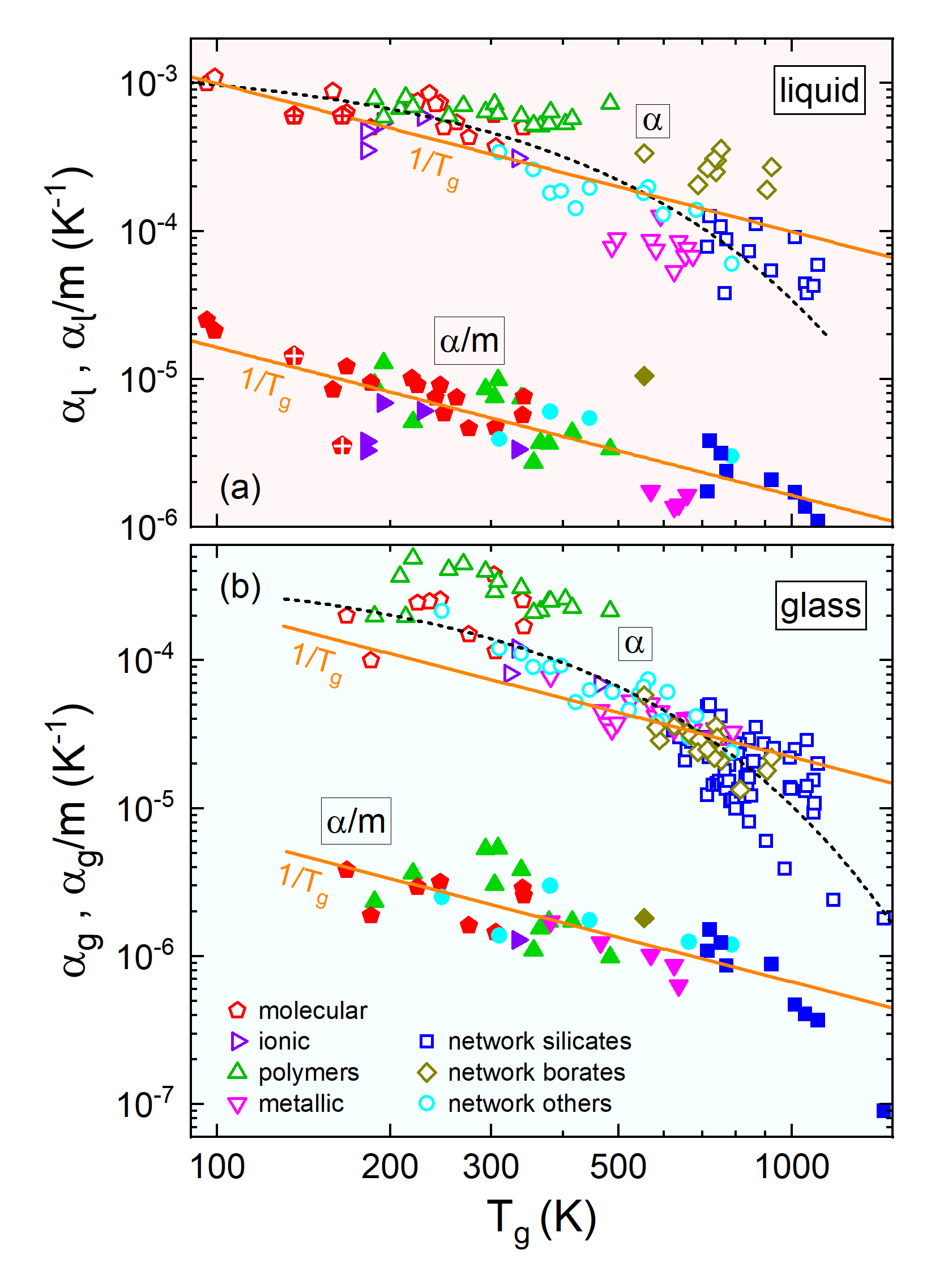
|
Fig. 2: Double-logarithmic plots of the experimentally-determined thermal volume-expansion coefficients αg
in the glass phase (b) and of αl in the liquid phase (a) versus the glass temperature Tg
for a large variety of glass formers belonging to different material classes. In addition to the bare expansion
coefficients (open symbols), the figure also provides the α values divided by the fragility parameter m
(filled symbols) being a measure of cooperative dynamics. The solid lines show linear fits with slope -1 corresponding
to 1/Tg dependence. The dashed lines indicate stronger Tg dependence for the bare α data.
Note that the ordinates of (a) and (b) were adjusted to achieve the same decades/cm ratio.
[from: P. Lunkenheimer, A. Loidl, B. Riechers, A. Zaccone, and K. Samwer, Thermal expansion and the glass transition, Nature Phys. 19, 694 (2023)]. |
At the glass transition, most physical quantities reveal a crossover to weaker temperature dependence. This is also the case for the volume (Fig. 3), implying that the thermal expansion of the liquid is larger than that of the solid glass. While vibrations contribute to the expansion in both solid and liquid, in the liquid there are additional configurational contributions (cf. Fig. 1).
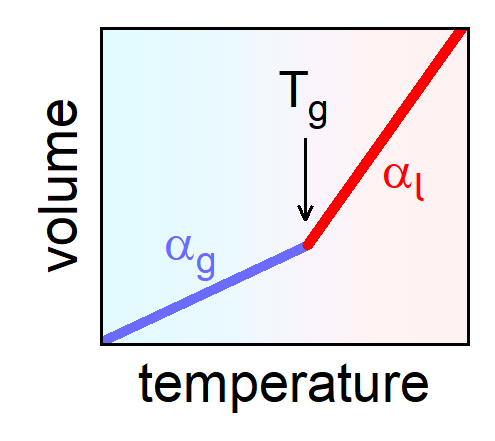
|
Fig. 3: Volume vs. temperature around the glass transition.
[from: P. Lunkenheimer, A. Loidl, B. Riechers, A. Zaccone, and K. Samwer, Thermal expansion and the glass transition, Nature Phys. 19, 694 (2023)]. |
The vast set of data collected in the framework of the present research reveals another surprisingly universal correlation: The thermal expansion in the liquid state is also correlated with the glass-transition temperature, in a very similar way as for the glass state (Fig. 2a). Moreover, we find that it is by a universal factor of about 3 larger than in the glassy state of a material, irrespective of the particular material class (except for the borates which exhibit anomalous expansion behaviour). This is surprising because the thermal expansion in both states of matter is commonly believed to be governed by fundamentally different mechanisms: Vibrations in the solid glass, in contrast to dominant translational motions in the liquid (see Fig. 1).
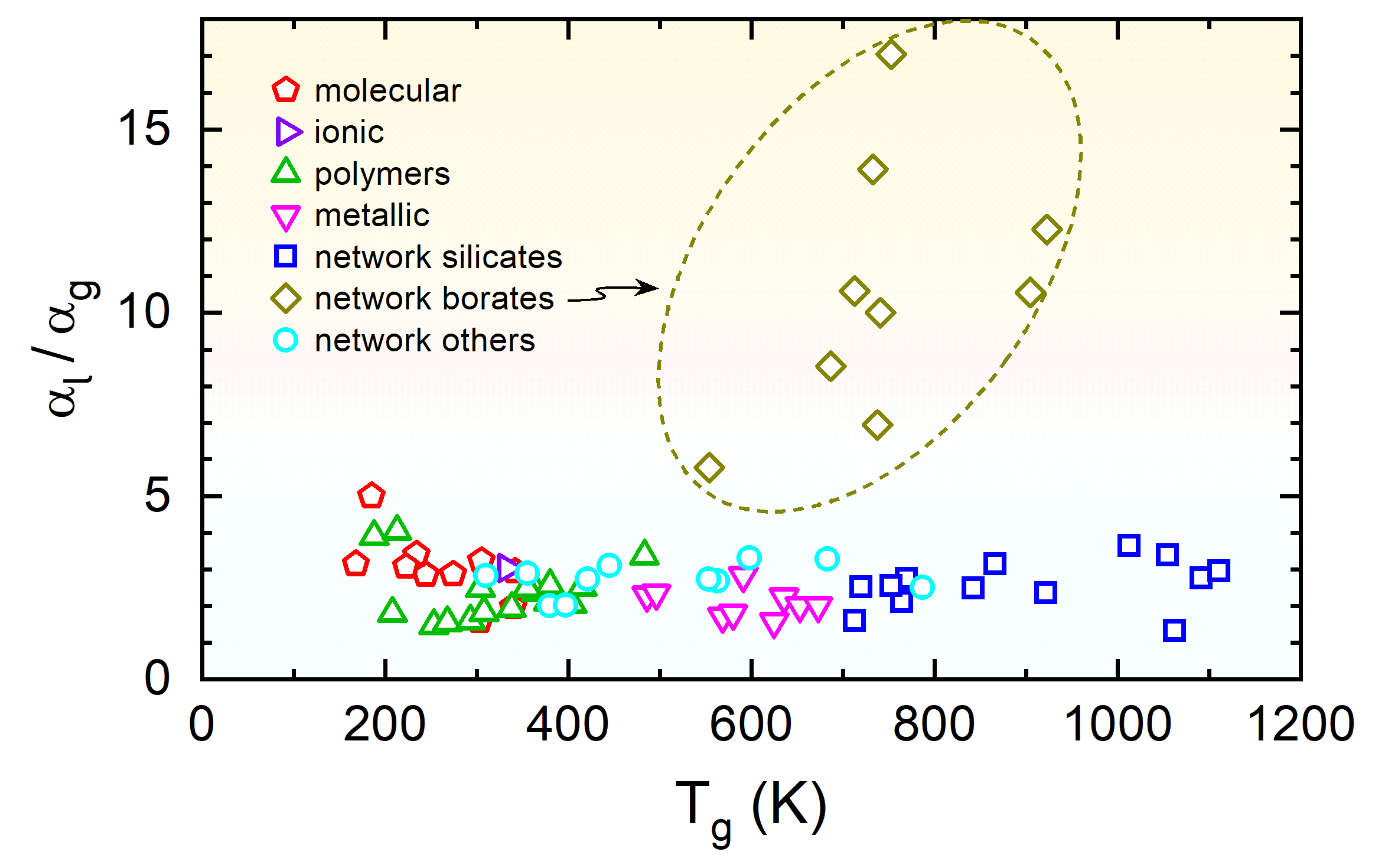
|
Fig. 4: Ratio of the thermal expansion coefficients measured in the liquid and glass phases.
Leaving the borate glasses aside, this ratio is essentially independent of the glass temperature
and of order three for all systems.
[from: P. Lunkenheimer, A. Loidl, B. Riechers, A. Zaccone, and K. Samwer, Thermal expansion and the glass transition, Nature Phys. 19, 694 (2023)]. |
In conclusion, the found universal correlation of thermal expansion and glass-transition temperature in different material classes, involving the degree of cooperativity of particle motion, obviously is a typical, so far unnoticed property of glasses. It markedly differs from the much simpler behaviour of crystalline systems which can be explained in terms of the Lindemann criterion. This and the unexpected universal factor relating α in the glass to that in the liquid put severe constraints on existing and future models of the glass transition.
3. Publications:
| [1] | Thermal expansion and the glass transition P. Lunkenheimer, A. Loidl, B. Riechers, A. Zaccone, and K. Samwer, Nature Phys. 19, 694 (2023). [PDF] |
| [2] | Universelle thermische Ausdehnung in Gläsern P. Lunkenheimer, A. Loidl, B. Riechers, und K. Samwer, Physik in unserer Zeit 54, 112 (2023). |